Ion exchange resin
Ion exchange resin
Types of ion exchange resins
Types of ion exchange resins include strong acidic cationic resins, weak acidic cations, strong anionic base, weak base anionic and chelaitng resins. Cationic and anionic resins are widely used in water treatment. Here, with each naming of these resins, we discuss their different types and then describe the equations.
Strong acidic cationic resins
One of the types of ion exchange resins is strong acidic cationic resins, naming these strong acidic resins because of their chemical behavior similar to that of strong acids. Resins are highly ionized both in acidic form (R-SO3H) and in salt form (R-SO3Na) throughout the ph range.
Poorly acidic cationic resins
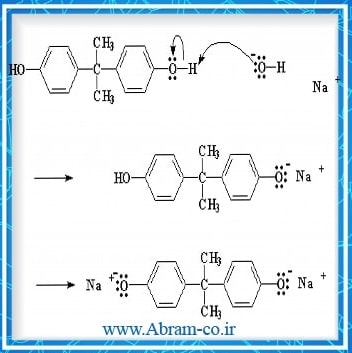
one of the types of ion exchange resins is weak acidic cationic resins which in the weak acid resins are carboxylic acid ionizable group (COOH-) whereas in the strong acidic resin of this group is a sulfonic acid (SO3H-) group. . These resins, such as weak organic acids, are degraded in small quantities.
Anionic resins strong match
Like strong acidic resins, strong play resins are also highly ionized and can be applied throughout the PH range. These resins are used in the form of hydroxide (OH) for deionization of water.
Poor anionic resins play
In weak resins such as low acid resins, the degree of ionization is strongly affected by PH.
Chelating Resins Heavy Metal Selector
Heat-chelating resins act as weak acidic cationic resins except that heavy metals tend to be heavily selective. Chelated resins have a strong tendency to form stable complexes with heavy metals. In fact, the functional groups used in these resins are EDTA compounds. The shape of the sodium resin structure is expressed as R-EDTA-Na.
The reactions occurring in a variety of ion exchange resins depend on the position of the chemical equilibria in which one ion is selectively substituted with another ion in place of the ionized exchange. Cation exchange on the sodium cycle can be demonstrated by the following reaction:
Na2.R + Ca+ <=> Ca+.R + Na+